
Sodium Perborate for Lead Acid Battery manufacturing as additive
Sodium Perborate is known as oxidant under acid-alkaline media to demonstrate the performance of a membraneless sodium perborate fuel cell (MLSPBFC). Sodium perborate is a true peroxygen salt and is a convenient source of hydrogen peroxide
The sodium perborate fuel cell (MLSPBFC) is unique from previous fuel cells using, and sodium perborate can be used not only as an oxidant but also as a reductant 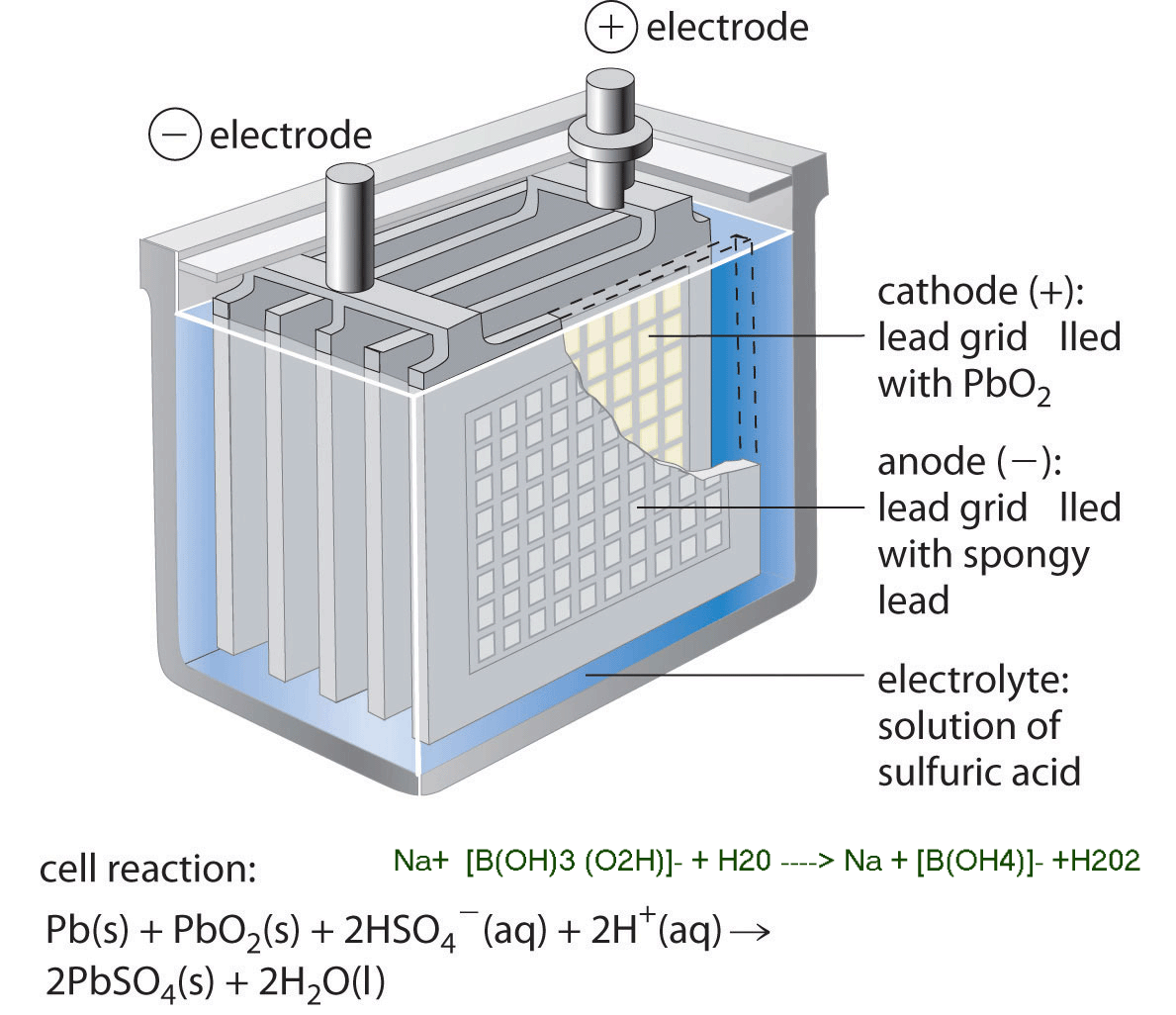
The MLSPBFC has some advantages like sodium perborate is a cheap, nontoxic, and large scale industrial chemical which is used primarily in detergents and as a mild oxidant. The cell being more environmentally friendly than the other fuel cells and the sodium perborate can be handled more simply than hydrogen peroxide, as it is a well-known fact that sodium perborate solution is a widespread safe disinfectant. On the performance side, the MLSPBFC generates electric power comparable to a typical air-breathing DMFC when operated in a microchemical channel at room temperature. In addition, the MLSPBFC requires no membrane electrode assemblies. Thus, the cost for the materials is low and the structure of the cell is simple.
Battery manufacturers may add other additive to the positive paste in order to to enhance features of the production process (e.g. to improve the paste-grid adhesion, reduce the formation time etc). Several of these additives have been show to influence the effect of lead acid battery on the crystal growth process.
The top manufacturer of lead acid manufacturer actually are using sodium perborate perborate to promote free lead reduction and to improve paste/grid adehnsion. In serveral trials the addition of 0.5% up to % sodium perborate tetrahydrate had beeen show to negative influence the battery and lead to a broader crystal size distribution as well as some larger agglomerates.
Using a small quantity of sodium perborate tetrahydrate in the positive plate mix which addition agent upon final curing of the plate with the paste therein causes the plate paste to be extremely hard and resistant to shatter.
Learn more about sodium perborate
Download
Specification Data Sheet [ Sodium Perborate Tetrahydrate ]
Material Safety Data Sheet [ Sodium Perborate Monohydrate ]
Material Safety Data Sheet [ Sodium Perborate Tetrahydrate ]
